Translate this page into:
Unmasking hyperandrogenism: What clinical symptoms reveal about hormonal mysteries

*Corresponding author: Palvi Singla Jindal, Department of Internal Medicine, Maimonides Medical Center, Brooklyn, United States. palvisingla7@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Jindal PS, Choudhary AN, Joshi RR, Shah B. Unmasking hyperandrogenism: What clinical symptoms reveal about hormonal mysteries. CosmoDerma. 2025;5:38. doi: 10.25259/CSDM_223_2024
Abstract
Objectives
This single-center observational study aimed to explore the relationship between clinical features and hormonal indices in affected patients while determining the underlying etiology of hyperandrogenism.
Materials and Methods
In this cross-sectional study performed at a tertiary care hospital, 200 patients with features of hyperandrogenism were included. Patients underwent comprehensive clinical assessment and laboratory tests, including serum hormonal profile, free androgen index (FAI), fasting blood glucose, fasting insulin, and Homeostatic Model Assessment of Insulin Resistance.
Results
The mean age of the participants was 28.80 ± 10.15 years (range 15–59 years). Hirsutism was the most prevalent symptom (99%) followed by alopecia (61.5%), acne (54.5%), seborrhea (38%), and acanthosis nigricans (29.5%). Polycystic ovary syndrome (PCOS) was the leading cause (60%) followed by idiopathic hirsutism (19.5%). Mean values of serum luteinizing hormone/follicle-stimulating hormone ratio, total and free testosterone, and FAI were elevated beyond the reference range in patients with clinical features of hyperandrogenism, suggesting a link between these clinical features and altered hormonal profile. However, despite these elevated levels, no significant correlation was found between specific clinical symptoms and hormonal levels.
Conclusion
The study identified ovarian causes as predominant, with significant associations between PCOS and elevated serum androgens. However, clinical manifestations of hyperandrogenism were not reliable predictors of specific hormonal abnormalities, suggesting that biochemical assessment is crucial for accurate diagnosis.
Keywords
Acne
Etiology of hyperandrogenism
Hirsutism
Hyperandrogenism
PCOS
INTRODUCTION
Hyperandrogenism, or androgen excess, is a medical condition characterized by excessive production and/or secretion of androgens by the ovaries and/or the adrenal glands where hirsutism, acne, and androgenetic alopecia (AGA) are some of the common clinical cutaneous manifestations. Hyperandrogenism is seen in around 7% of the reproductive age group females.[1] Hirsutism, acne, and AGA may be seen in isolation or can present as part of an underlying hyperandrogenic state in 30–50% of patients.[2] It was Bullock and Sequeira who in 1905 first brought up the association between masculinisation and endocrine pathology.[3]
These manifestations are fairly common, where hirsutism is seen in around 60–83%, acne in 11–43%, and AGA in 25% of females.[1,4,5] Other clinical manifestations of hyperandrogenism include weight gain, acanthosis nigricans, striae, menstrual irregularity, infertility, virilization, and pelvic mass. There is an increase in hirsutism with age due to more duration of exposure to androgens.[6]
Cutaneous hyperandrogenism can signify an idiopathic skin disease or a systemic disorder like polycystic ovary syndrome (PCOS), the most commonly diagnosed underlying condition in about 80–85% of reproductive age women.[7] Other causes include non-classic congenital adrenal hyperplasia, androgen-secreting tumors, Cushing syndrome, thyroid dysfunction, hyperprolactinemia, and the use of specific drugs (e.g., testosterone, dehydroepiandrosterone sulfate, danazol, corticotropin, high-dose corticosteroids, metyrapone, phenothiazine derivatives, anabolic steroids, androgenic progestin, and acetazolamide).[7-9] Idiopathic hirsutism (5–20% of cases) is characterized by regular menses without androgen excess.[10,11] It can be due to disorders in peripheral androgen activity and is often associated with a positive family history.[12]
Among the various disorders presenting with cutaneous hyperandrogenism, history including age, onset of symptoms, chronology, and progression helps to identify likely diagnoses. For example, females <35 years of age with slow hair growth and the presence of menstrual irregularities shortly after puberty have a high possibility of PCOS[13] whereas ones more than 35 years, with rapid excessive hair growth, deepening of the voice, and breast atrophy would more likely be a case of adrenal or ovarian tumor.
The purpose of this article was to review the occurrence of cutaneous manifestations, laboratory abnormalities, underlying etiopathology, and their relative frequency, along with finding an association among severity of clinical features and levels of androgenic hormones if any in patients with hyperandrogenism.
MATERIALS AND METHODS
In this cross-sectional study, 200 female patients aged 15–60 years with clinical signs of hyperandrogenism from the dermatology department of a tertiary care hospital in India from August 2016 to October 2018 were included in the study.
Inclusion criteria
Presence of clinical signs or symptoms of hyperandrogenism (e.g., hirsutism, virilization, menstrual irregularities, or androgenic alopecia).
Exclusion criteria
Pregnant or breastfeeding individuals, as hormonal fluctuations, may confound the results.
Use of medications known to influence androgen levels (e.g., corticosteroids, oral contraceptives, or antiandrogens) within 3 months before enrollment.
After informed consent from the patients, participants underwent detailed clinical evaluations using a standardized proforma to collect data on age, symptom onset, menstrual history, family history of related conditions, treatment history, and symptoms of virilization such as deepening of the voice, frontal balding, seborrhea, increased muscularity, and decreased breast size. Menstrual cycles were categorized as regular or irregular based on cycle length.
Dermatological assessments included evaluating hirsutism using the modified Ferriman–Gallwey (mFG) method, acne severity, hair loss, seborrhea, and other signs of androgen excess. Clitoromegaly was noted if the size exceeded 35 mm2. Other endocrinopathy signs were also recorded.
Metabolic assessments included height, weight, body mass index (BMI), waist and hip circumference, waist–to–hip ratio (WHR), and blood pressure. Abnormal WHR was defined as WHR ≥0.85 and obesity as BMI ≥30 kg/m2.
Laboratory tests included serum hormonal profile [thyroid hormonal profile (thyroid function tests), luteinizing hormone (LH), follicle-stimulating hormone (FSH), total and free testosterone, prolactin, dehydroepiandrosterone sulfate (DHEAS), sex hormone binding globulin, 17-hydroxyprogesterone (17-OHP)] and calculated free androgen index (FAI). Metabolic syndrome was assessed through fasting blood glucose, insulin levels, lipid profile, and Homeostatic Model Assessment of Insulin Resistance. Abdominal and pelvic ultrasound on days 3–5 of the menstrual cycle identified polycystic ovaries by detecting ≥12 follicles or ≥10 mL ovarian volume.[14]
Statistical analysis
The data were entered into a spreadsheet and then described in terms of range, mean ±standard deviation, frequencies (number of cases), and relative frequencies (percentages) as appropriate. For comparing categorical data, a Chi-square (c2) test was performed and a probability value (P-value) <0.05 was considered statistically significant. All statistical calculations were done using the Statistical Package for the Social Science version 21.
RESULTS
Demographics
The study encompassed 200 women with a mean age of 28.80 ± 10.15 years (range 15–59 years) predominantly in the 20–29-year age group (101/200; 50.5%). The average symptom duration onset before presentation was 4.14 ± 2.84 years (range 1–20 years) [Table 1].
| Characteristics | Hyperandrogenic patients |
|---|---|
| Age (years) | |
| Mean age | 28.50±10.15 |
| Range | 15–59 |
| Age groups (years) (%) | |
| 10–19 | 28 (14) |
| 20–29 | 101 (50.5) |
| 30–39 | 41 (20.5) |
| 40–49 | 16 (8) |
| 50–59 | 14 (7) |
| Symptom onset (years) | |
| Mean duration | 4.14±2.84 |
| Range | 1–20 |
| Family history (%) | |
| Present | 31 (15.5) |
| Marital history (%) | |
| Unmarried | 114 (57) |
| Married | 86 (43) |
Menstrual and family history
Irregular menstruation was reported by 90/200 (45%) patients, with 70/200 (35%) experiencing oligomenorrhea and 20/200 (10%) menorrhagia. Fourteen/200 (7%) patients were menopausal. Among the 70/200 patients with oligomenorrhea, 54/200 (27%) had PCOS as the underlying cause.
A family history of hyperandrogenism was present in 31/200 (15.5%) patients with a family history of PCOS in 15/200 (48.3%) patients. Discrete hirsutism (17/200; 55%) was more common in patients with positive family history [Table 1].
Marital and obstetric history
Unmarried women (114/200; 57%) were more common than the married ones (86/200; 43%). Infertility was noted in 18/200 (20.9%) married women, with PCOS accounting for 83.3% of these cases [Tables 1 and 2].
| S. No. | Clinical features | Number of patients (%age) |
|---|---|---|
| 1. | Hirsutism (mFG score) | |
| Discrete (8–16) | 85 (42.5) | |
| Moderate (17–25) | 96 (48) | |
| Significant (>25) | 17 (8.5) | |
| 2. | Alopecia (pattern) | |
| Diffuse hair loss | 72 (58.5) | |
| MPHL | 27 (21.9) | |
| FPHL | 24 (19.5) | |
| 3. | Acne | |
| Grade 1 | 15 (13.7) | |
| Grade 2 | 45 (41.2) | |
| Grade 3 | 41 (37.6) | |
| Grade 4 | 8 (7.3) | |
| 4. | Seborrhea | 76 (38) |
| 5. | Acanthosis nigricans | 59 (29.5) |
| 6. | Menstrual abnormalities | |
| Oligomenorrhea | 70 (35) | |
| Menorrhagia | 20 (10) | |
| 7. | Infertility | 18 (20.9) |
| 8. | Obesity (BMI >25 kg/m2) | |
| Pre-obese (25–30) | 78 (39) | |
| Obese (30–37) | 10 (5) | |
| Morbid obesity (>37) | 6 (3) | |
| 9. | Waist–hip ratio (>0.85) | 88 (44) |
| 10. | Striae | 7 (3.5) |
| 11. | Increase muscle mass | 6 (3) |
| 12. | Deepening of voice | 6 (3) |
| 13. | Clitoromegaly | 4 (2) |
mFG: modified Ferriman Gallawey score, MPHL: Male pattern hair loss, FPHL: Female pattern hair loss, BMI: Body mass index
Symptomatology
Among features of hyperandrogenism, hirsutism was the most prevalent symptom (198/200; 99%), followed by alopecia (123/200; 61.5%), acne (109/200; 54.5%), seborrhea (76/200; 38%), and acanthosis nigricans (59/200; 29.5%). Hirsutism was severe in 17/200 (8.5%) cases, with moderate and mild cases in 96/200 (48%) and 85/200 (42.5%), respectively. Acne was most commonly grade 2 (45/200; 22.5%) and grade 3 (41/200; 20.5%) [Table 2].
Examination findings
The mean BMI was 24.68 ± 4.77 kg/m2 (range 17.8–41.6 kg/m2) with 94/200 (47%) females categorized as overweight or obese. A significant proportion (79/200; 38.5%) had an elevated WHR [Table 2].
Laboratory findings
Mean values of serum LH/FSH ratio, total testosterone, free testosterone, and FAI were elevated beyond the reference range in patients with clinical hyperandrogenism [Table 3]. Notably, PCOS patients frequently exhibited high levels of these hormones. Elevated DHEAS and prolactin were observed in specific cases, with ovarian tumors showing the highest levels of testosterone and DHEAS [Table 4].
| Clinical features | Investigations | ||||
|---|---|---|---|---|---|
| LH/FSH | DHEAS (μg/dL) | Total testosterone (ng/dL) | Free testosterone (ng/dL) | Free androgen index | |
| Hirsutism | 2.57±1.29 | 308±69 | 94.31±52.15 | 2.30±9.09 | 12.32±8.02 |
| Acne | 2.67±1.33 | 306.05±147.01 | 93.70±43.71 | 1.73±0.99 | 13.31±8.56 |
| Alopecia | 2.63±1.36 | 308.74±153.30 | 91.95±40.32 | 2.71±11.51 | 12.42±7.90 |
| Acanthosis nigricans | 3.17±1.30 | 347.43±202.43 | 111.53±71.20 | 1.81±1.10 | 15.94±9.78 |
| Oligomenorrhea | 3.02±1.32 | 347.24±204.38 | 112.23±63.33 | 1.74±0.90 | 15.60±8.75 |
LH: Luteinizing hormone, FSH: Follicle stimulating hormone, DHEAS: Dehydroepiandrosterone-sulfate
| S. No. | Investigations | Number of patients (% age) | |
|---|---|---|---|
| 1. | LH/FSH ratio (≥2:1) | 123 (62.5) | |
| 2. | Prolactin (>29.93 ng/mL) | 6 (3) | |
| 3. | DHEAS (>430 mcg/dL) | 47 (23.5) | |
| 4. | 17-OHP (>30 mmol/L) | 19 (9.5) | |
| 5. | Total testosterone (>60 ng/dL) | 142 (71) | |
| 6. | Free testosterone (>1.3 ng/dL) | 94 (47) | |
| 7. | SHBG (<18 nmol/L) | 34 (17) | |
| 7. | Free androgen index (>10) | 90 (45) | |
| 8. | FBS (>110 mg/dL) | 66 (33) | |
| 9. | PPBS (>140 mg/dL) | 49 (24.5) | |
| 10. | Fasting insulin (>17 Miu/mL) | 32 (16) | |
| 11. | HOMA-IR (>3) | 88 (44) | |
| 12. | TSH | 12 (6) | |
| 13. | Altered lipid profile | 19 (9.5) | |
| 14. | USG findings | ||
| Polycystic ovaries | 115 (57.5) | ||
| Solid tumor in ovaries | 2 (1) | ||
LH: Luteinizing hormone, FSH: Follicle-stimulating hormone, DHEAS: Dehydroepiandrosterone-sulfate, 17-OHP: 17-hydroxyprogesterone, FBS: Fasting blood glucose, HOMA-IR: Homeostatic model assessment of Insulin resistance, TSH: Thyroid-stimulating hormone, USG: Ultrasonography, SHBG: Sex hormone binding globulin, PPBS: Postprandial blood sugar
Table 5 summarizes the comparison of clinical and hormonal features by underlying clinicopathological conditions.
| PCOS | Idiopathic | Familial | Hyperprolactinemia | |
|---|---|---|---|---|
| Mean age (years) (range) | 27.89±10.45 (15–59) | 29.35±9.47 (17–54) | 29.18±9.86 (17–52) | 27.25±5.5 (19–30) |
| Mean BMI (kg/m2) | 24.75±4.71 | 26.67±4.69 | 24.03±2.44 | 24.04±3.96 |
| Mean WHR | 0.88±0.06 | 0.84±0.05 | 0.84±0.05 | 0.86±0.04 |
| Mean mFG | 17±6.0 | 16.15±6.60 | 16.43±7.44 | 19.5±5.44 |
| LH/FSH | 3.5±0.79 | 1.27±0.38 | 1.24±0.23 | 1.30±0.21 |
| Total testosterone (ng/dL) | 112.2±1.01 | 55.79±11.81 | 65.56±7.88 | 51.25±10.6 |
| Free testosterone (pg/mL) | 1.97±8.27 | 1.10±0.36 | 1.18±0.07 | 0.93±0.04 |
| Free androgen index | 15.78±8.27 | 6.52±1.36 | 5.55±1.40 | 7.57±0.85 |
| DHEAS (μg/dL) | 324.69±137.60 | 162.48±35.69 | 191.11±53.45 | 516.47±130.0 |
| Estradiol (pg/mL) | 97.93±73.07 | 89.56±66.34 | 70.26±59.46 | 46.91±13.51 |
| Prolactin (ng/mL) | 15.4±7.08 | 15.54±6.25 | 12.53±2.21 | 207.5±53.20 |
| FBS (mg/dL) | 113.72±26.25 | 93.10±8.38 | 104.75±14.41 | 99±7.39 |
| Fasting insulin (mIU/L) | 12.78±6.13 | 9.67±3.91 | 11.46±4.0 | 9.47±1.73 |
| HOMA-IR | 3.81±2.66 | 2.23±0.95 | 3.10±1.58 | 2.30±0.41 |
| Hypothyroidism | Adrenal hyperandrogenism | Cushing syndrome | Androgen-secreting ovarian tumor | |
| Mean age (years) (range) | 30.75±4.5 (27–36) | 29.84±9.20 (18–46) | 42±8.48 (36–48) | 49±9.8 (42–56) |
| Mean BMI (kg/m2) | 26.73±8.38 | 25.09±5.31 | 36.16±2.87 | 28.57±5.12 |
| Mean WHR | 0.86±0.06 | 0.86±0.06 | 0.91±0.02 | 0.83±0.02 |
| Mean mFG | 9.5±0.57 | 19.69±5.49 | 23.5±3.53 | 31±1.41 |
| LH/FSH | 0.82±0.07 | 1.13±0.56 | 1.86±0.94 | 1.04±0.36 |
| Total testosterone (ng/dL) | 72±6.9 | 52.23±16.58 | 73.5±9.19 | 400.5±28.99 |
| Free testosterone (pg/mL) | 1.36±0.04 | 1.27±0.31 | 0.81±0.70 | 6.06±3.95 |
| Free androgen index | 6.9±0.69 | 7.61±2.06 | 10.23±3.20 | 26.48±0.57 |
| DHEAS (μg/dL) | 406±50.95 | 504.76±41.83 | 456.35±64.13 | 1149.4±228.25 |
| Estradiol (pg/mL) | 92±8.08 | 115.73±81.42 | 38.68±8.93 | 53.22±19.48 |
| Prolactin (ng/mL) | 11.74±1.91 | 13.46±5.67 | 6.32±1.01 | 7.9±0.42 |
| FBS (mg/dL) | 98.25±3.30 | 104.39±26.40 | 129±14.14 | 106±25.45 |
| Fasting insulin (mIU/L) | 12.97±0.03 | 13.76±9.53 | 23.5±6.36 | 14±2.82 |
| HOMA-IR | 3.13±0.10 | 4.22±5.28 | 7.59±2.84 | 3.75±1.62 |
PCOS: Polycystic ovary syndrome, BMI: Body mass index, WHR: Waist-hip ratio, m-FG: Modified Ferriman-Gallwey score, LH: Luteinizing hormone, FSH: Follicle-stimulating hormone, DHEAS: Dehydroepiandrosterone-sulfate, FBS: Fasting blood glucose, HOMA-IR: Homeostatic model assessment of Insulin Resistance
Ultrasound findings
Ovarian changes suggestive of PCOS were found in 115/200 (57.5%) patients while solid ovarian tumors were noticed in 2/200 (1%) patients.
Etiology
In this study, PCOS [Figure 1] was the leading diagnosis, affecting 120/200 (60%) patients, followed by idiopathic hirsutism (39/200; 19.5%), familial (16/200; 8%), and adrenal (13/200; 6.5%). Hypothyroidism and hyperprolactinemia were less common (4/200; 2% each); 2/200 (1%) each had ovarian androgen-secreting neoplasm [Figure 2] and Cushing syndrome [Figure 3] affecting 4/200 (2%) patients [Table 6].
| Etiology | Malik et al., 2007[16] | Ansarin et al., 2007[11] | Ahmad et al., 2009[15] | Chhabra et al., 2012[21] | D Sharma et al., 2012[17] | Pathania et al., 2020[20] | Dhanotia et al., 2020[19] | Mahajan et al., 2020[18] | Present Study |
|---|---|---|---|---|---|---|---|---|---|
| PCOS | 44.6% | 62.53% | 11.43% | 70% | 38% | 72% | 38.64% | 29.5% | 60% |
| Idiopathic hirsutism | 47.3% | 35.19% | 80% | 15% | 50% | 13% | 43.18% | 10.7% | 19.5% |
| Familial | - | - | - | - | - | - | - | 1.6% | 8% |
| Adrenal hyperandrogenism | 1.3% | 0.38% | 2.86% | 7.5% | 4% | - | - | - | 6.5% |
| Hypothyroidism | 4% | - | 5.70% | 7.5% | 8% | 3% | - | - | 2% |
| Hyperprolactinemia | - | 0.13% | - | - | - | 1% | - | 10.7% | 2% |
| Ovarian androgen-secreting neoplasm | - | - | - | - | - | - | - | - | 1% |
| Cushing disease/syndrome | 2.7% | - | - | - | - | - | - | - | 1% |
| Iatrogenic | - | - | - | - | - | 4% | - | - | - |
PCOS: Polycystic ovary syndrome
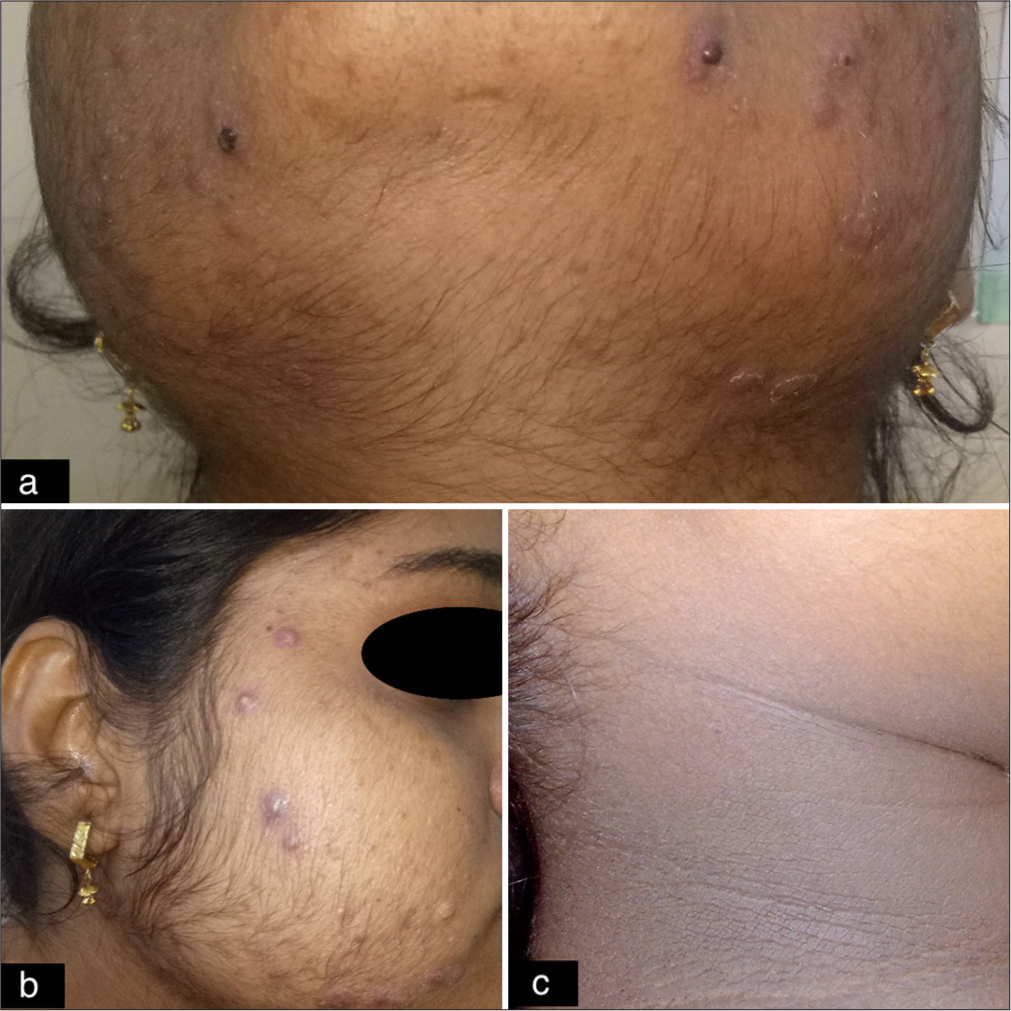
- Cutaneous findings found in PCOS females. (a) Hirsutism. (b) Acne vulgaris (grade III). (c) Acanthosis nigricans. PCOS: Polycystic ovary syndrome.
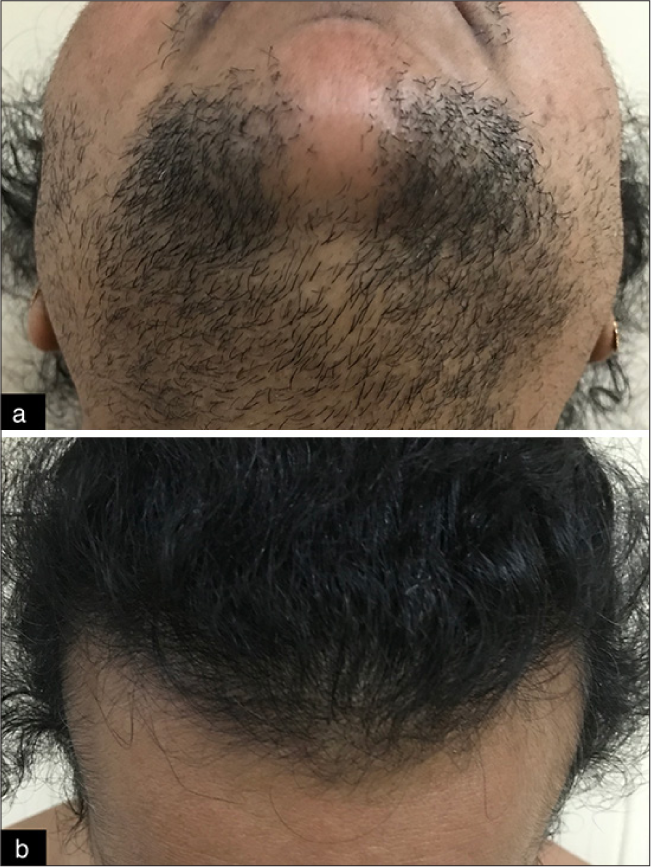
- Cutaneous features seen in patients with ovarian androgen-secreting tumors. (a) Severe hirsutism with m-FG of 31. (b) Male-pattern androgenetic alopecia (grade-III) in the patient.
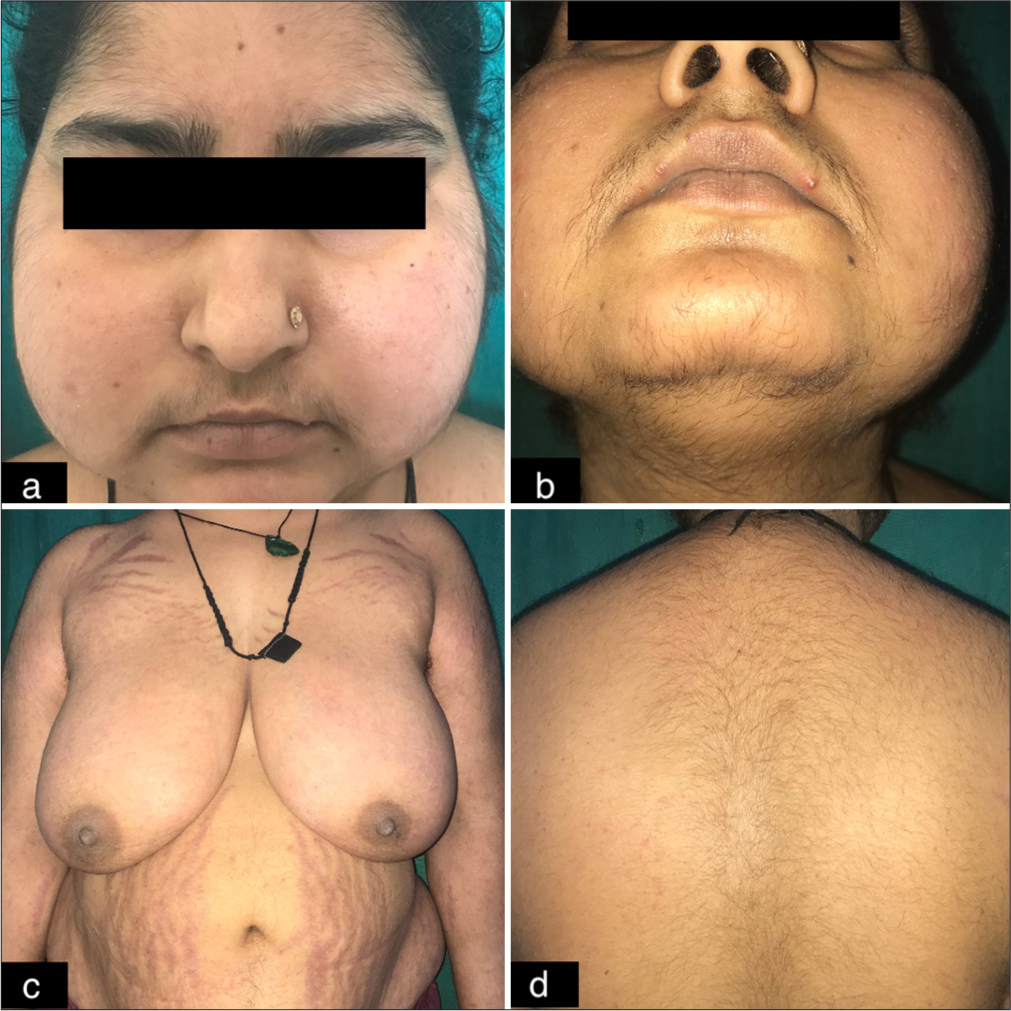
- Cutaneous findings in the patient with Cushing syndrome due to pituitary adenoma. (a) Cushingoid facies of the patient. (b) Hirsutism. (c) Purple striae over the shoulder, upper chest, and abdomen. (d) Buffalo hump.
Comparison of PCOS and non-PCOS groups
Significant differences were observed in the occurrence of oligomenorrhea, infertility, acanthosis nigricans, WHR, BMI, LH/FSH ratio, total and free testosterone, FAI, fasting blood sugar, and fasting insulin levels between PCOS and non-PCOS groups. It indicates that PCOS is not only an infertility problem or a cosmetic annoyance but also it is foremost a general health problem. Therefore, any therapy for PCOS should optimally target not only the ovulatory dysfunction and hyperandrogenism but also the co-morbidities associated with it.
DISCUSSION
This study provides a comprehensive overview of cutaneous manifestations associated with hyperandrogenism, exploring their occurrence, laboratory abnormalities, and underlying etiologies.
The majority of our patients were aged 20–29 years, aligning with previous research indicating that this is a common age for hirsutism onset.[7-9,12,15-22] The higher frequency of unmarried women in our cohort may reflect cultural or societal influences as seen in other studies.[17,18,21] Patients typically sought help after an average of 4.14 years, consistent with reported durations in similar studies.[19,21] A family history of hyperandrogenism was present in 15.5% of cases, echoing previous findings[9,21] but with some variation.[15,18]
Hirsutism was observed in 99% of patients, with a mean mFG score of 17.05 ± 6.37, comparable to some studies[16,18] but higher than others.[7,9,20] Notably, severe hirsutism was observed in 8.5% of the cases, aligning with findings that link severe symptoms to higher androgen levels. Alopecia was noted in 61.5% of patients, with AGA affecting 13.5%. Acne and oligomenorrhea were prevalent in 54.5% and 35% of patients, respectively. A significant association between menstrual irregularities and PCOS was found, aligning with other studies.[17,21] This is in line with the association between menstrual irregularities and hyperandrogenic states. Infertility, reported in 20.9% of married women, underscores the reproductive impact of hyperandrogenism, with PCOS being the predominant cause.
Hormonal profiles revealed elevated LH/FSH ratios in 62.5% of patients, with most having PCOS which aligns with several studies.[16,18,21,23] Elevated serum total testosterone was seen in 71% of patients, consistent with several studies,[16,20] though variation exists.[15,19] Serum-free testosterone was elevated in 47% of patients, with a significant association with PCOS. Elevated total testosterone and free testosterone levels were notably higher in patients with PCOS, while ovarian tumors exhibited the highest levels of these hormones. This highlights the importance of differentiating between PCOS and other conditions such as adrenal or ovarian tumors based on hormonal profiles. No significant correlation was found between the severity of hirsutism and free testosterone levels in our study, aligning with others,[24] but some found a significant association between them.[18,21,25] Similarly, no significant association was found between the severity of hirsutism and total testosterone and DHEAS levels in the present study, similar to others[18,21,24,25] while Pathania et al.,[20] found a significant association between raised serum total testosterone and severity of hirsutism. In addition, other clinical features such as hyperandrogenism and obesity showed no significant correlation with hormonal levels, consistent with Pathania et al.[20]
Interestingly, elevated serum DHEAS was noted in 23.5% of patients, with the highest levels observed in ovarian tumor cases. In ovarian tumors, particularly androgen-secreting tumors like Sertoli-Leydig cell tumors, the elevation of DHEAS may occur alongside other androgens such as testosterone. While DHEAS elevation is more commonly associated with adrenal causes, the presence of high levels in ovarian tumors can provide diagnostic insight, especially when considered alongside clinical features such as virilization, hirsutism, or menstrual irregularities. Thus, DHEAS measurement, while not diagnostic on its own, can aid in distinguishing between adrenal and ovarian sources of hyperandrogenism when combined with imaging and other hormone evaluations.
Biochemical hyperandrogenemia was found in 67.5% of patients, with a higher frequency in those with PCOS (8.75%) compared to non-PCOS patients. These findings are consistent with Chhabra et al.,[21] who reported 71.43% of PCOS patients with hyperandrogenemia, and Steinberger et al., who also observed significantly elevated androgen levels in PCOS patients.[26]
PCOS was the most common cause of hirsutism, present in 60% of patients, with idiopathic hirsutism and familial cases being the next most frequent. Adrenal causes were identified in 6.5% of patients, including both benign and malignant conditions. We observed elevated androgen and 17-OHP levels in patients with late-onset congenital adrenal hyperplasia. Clitoromegaly was noted in three patients with adrenal hyperandrogenism, and one patient with clitoromegaly had an ovarian androgen-secreting tumor. According to literature, these androgen-secreting tumors are relatively rare affecting one per 300–1,000 hirsute patients. Virilizing ovarian tumors constitute <0.2% of cases of hyperandrogenism.[27] This higher incidence of ovarian tumors in our study may be influenced by the referral of patients from our cancer institute, potentially skewing incidence figures.
Hypothyroidism and hyperprolactinemia were less common, and Cushing syndrome was present in 1% of cases. Table 6 illustrates the comparative underlying etiology of hyperandrogenism in several studies.
Overall, our study underscores the importance of a comprehensive evaluation in patients with hyperandrogenism, considering a range of potential causes and associated hormonal abnormalities.
Limitations
Limitations in this study can be a lack of a control group and no long-term follow-up, along with interobserver variations of dermatological examination, i.e., grading for acne and alopecia. Among investigations, anti-Müllerian hormone for PCOS diagnosis was not performed due to a lack of facilities in the center. Another limitation was that although patients were treated, the follow-up for therapeutic outcomes was not a part of the study.
CONCLUSION
This study underscores the importance of integrating hormonal evaluations with clinical observations to differentiate between physiological and pathological hyperandrogenism. Dermatologists often encounter the cutaneous manifestations of hyperandrogenism, positioning them as key players in its early detection and management. Despite the clinical and biochemical markers of hyperandrogenism often presenting in isolation, a thorough hormonal workup remains crucial for accurate diagnosis and tailored treatment. Moreover, it must be bear in mind that hyperandrogenemia is more detrimental to women’s health than clinical hyperandrogenism making it necessary to perform hormonal investigations for susceptible patients. Given that facial hair remains a primary concern for patients seeking dermatological care, our findings highlight the need for dermatologists to adopt a proactive approach to investigating underlying causes and managing associated metabolic abnormalities. While this study provides valuable insights, further research with larger samples and more refined methodologies is essential to enhance our understanding and management of hyperandrogenism.
Ethical approval
The research/study was approved by the Institutional Review Board at Institutional Ethics Committee/BJMC and Civil Hospital, Ahmedabad, number Ref no. IEC/Certi/91/17, dated May 5, 2017.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship: Nil.
References
- Polycystic ovary syndrome and the differential diagnosis of hyperandrogenism. Obstetr Gynaecol. 2013;15:171-6.
- [CrossRef] [Google Scholar]
- Evidence-based approach to cutaneous hyperandrogenism in women. J Am Acad Dermatol. 2015;73:672-90.
- [CrossRef] [PubMed] [Google Scholar]
- The relation of the suprarenal capsules to the sexual organs. Trans Path Soc Lond. 1905;56:189-208.
- [Google Scholar]
- Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88:4116-23.
- [CrossRef] [PubMed] [Google Scholar]
- Acne and hirsutism in polycystic ovary syndrome: Clinical, endocrine-metabolic and ultrasonographic differences. Gynecol Endocrinol. 2002;16:275-84.
- [CrossRef] [PubMed] [Google Scholar]
- Polycystic ovary syndrome in the adolescent. Obstet Gynecol Clin North Am. 2009;36:129-52.
- [CrossRef] [PubMed] [Google Scholar]
- Androgen excess in women: Experience with over 1000 consecutive patients. J Clin Endocrinol Metab. 2004;89:453-62.
- [CrossRef] [PubMed] [Google Scholar]
- Extensive clinical experience: Relative prevalence of different androgen excess disorders in 950 women referred because of clinical hyperandrogenism. J Clin Endocrinol Metab. 2006;91:2-6.
- [CrossRef] [PubMed] [Google Scholar]
- Hirsutism: Clinico-investigative profile of 50 Indian patients. Indian J Dermatol. 2008;53:111-4.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical presentation and etiologic factors of hirsutism in premenopausal Iranian women. Arch Iran Med. 2007;10:7-13.
- [Google Scholar]
- Hirsutism: Evaluation and treatment. Indian J Dermatol. 2010;55:3-7.
- [CrossRef] [PubMed] [Google Scholar]
- Phenotypic spectrum of polycystic ovary syndrome: Clinical and biochemical characterization of the three major clinical subgroups. Fertil Steril. 2005;83:1717-23.
- [CrossRef] [PubMed] [Google Scholar]
- Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS) Hum Reprod. 2004;19:41-7.
- [CrossRef] [PubMed] [Google Scholar]
- Hirsutism in Kashmir: An etiological study. Indian J Dermatol. 2009;54:80-2.
- [CrossRef] [PubMed] [Google Scholar]
- Clinico-investigative profile of patients of hirsutism in a tertiary level institution. Int J Trichology. 2012;4:69-74.
- [CrossRef] [PubMed] [Google Scholar]
- Clinico-investigative attributes of 122 patients with hirsutism: A 5-year retrospective study from India. Int J Womens Dermatol. 2020;7:237-42.
- [CrossRef] [PubMed] [Google Scholar]
- A study of clinical and investigational profile of hirsute women at a tertiary care center in Western India. Clin Dermatol Rev. 2020;4:123.
- [CrossRef] [Google Scholar]
- Clinico-pathological correlation in Indian women presenting with cutaneous features of hyperandrogenism: A cross-sectional study. Med J Dr. DY Patil Vidyapeeth. 2020;13:125.
- [CrossRef] [Google Scholar]
- Hirsutism: A Clinico-investigative study. Int J Trichology. 2012;4:246-50.
- [CrossRef] [PubMed] [Google Scholar]
- Correlation of biochemical markers and clinical signs of hyperandrogenism in women with polycystic ovary syndrome (PCOS) and women with non-classic congenital adrenal hyperplasia (NCAH) Iran J Reprod Med. 2012;10:307-14.
- [Google Scholar]
- Women with clinical significant hirsutism always have detectable endocrinological abnormalities. J Eur Acad Dermatol Venereol. 1997;9:226-31.
- [CrossRef] [Google Scholar]
- The prevalence of androgen excess among patients with minimal unwanted hair growth. Am J Obstet Gynecol. 2004;191:1914-20.
- [CrossRef] [PubMed] [Google Scholar]
- The grade of hirsutism correlated to serum androgen levels and hormonal indices. Acta Obstet Gynecol Scand. 1985;64:629-33.
- [CrossRef] [PubMed] [Google Scholar]
- Testosterone, dehydroepiandrosterone, and dehydroepiandrosterone sulfate in hyperandrogenic women. J Clin Endocrinol Metab. 1984;59:471-7.
- [CrossRef] [PubMed] [Google Scholar]






